Play/span>
Play/span>Video
Since 2003, Theirworld has invested £5 million in a research programme at the Jennifer Brown Research Laboratory (JBRL) in the Centre for Reproductive Health (CRH) at the University of Edinburgh. The research laboratory works to improve understanding of what causes early labour, how treatments can be developed to prevent it and how newborn babies can be better supported in the first crucial hours and days after birth.
Theirworld’s investment in research has contributed to establishing Edinburgh as a leading centre for perinatal experimental medicine research in the UK. Professor Boardman’s groundbreaking research from the Jennifer Brown Research Laboratory means the project could soon reach its potential internationally.
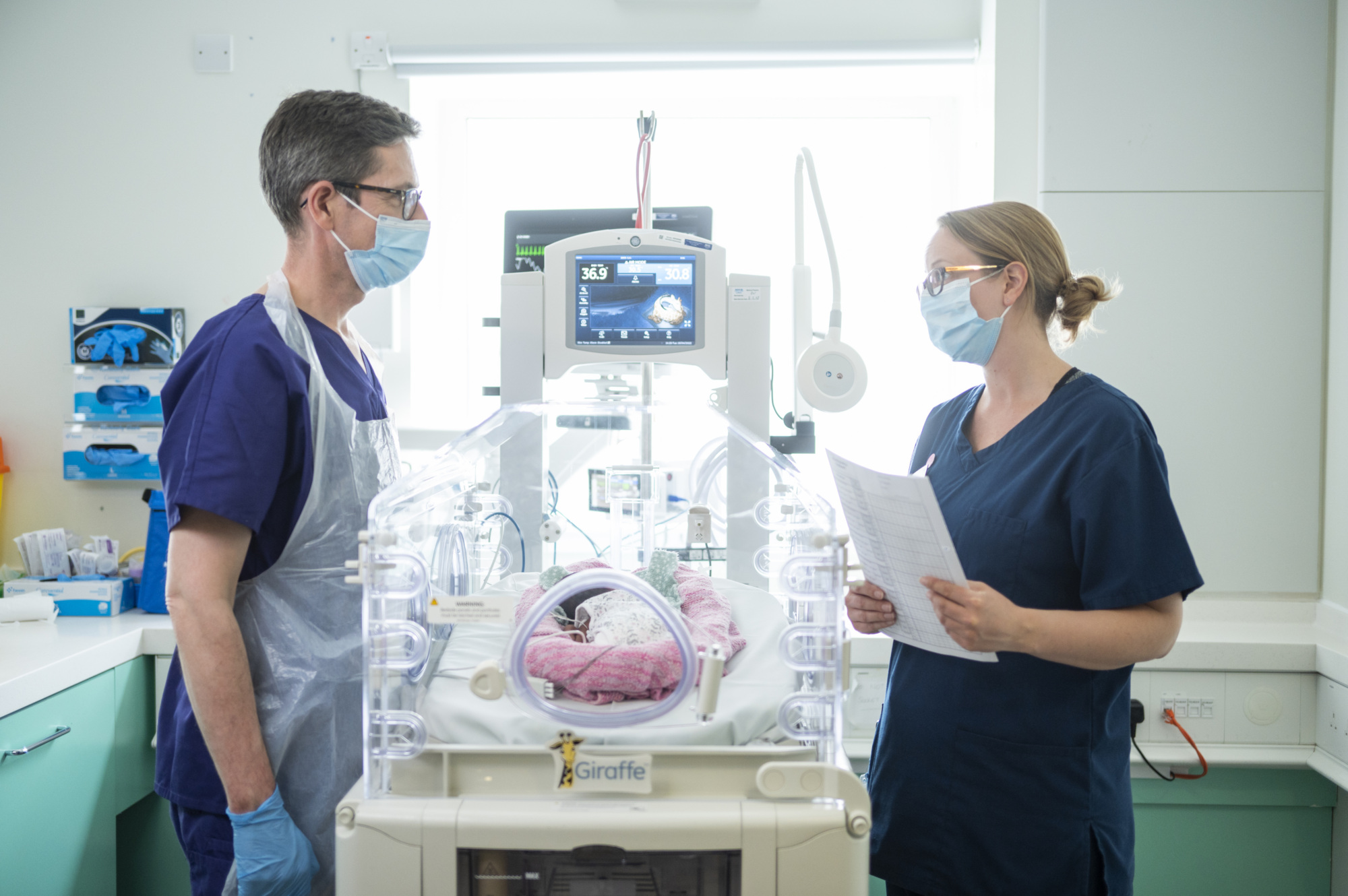
The laboratory’s flagship project is the Theirworld Edinburgh Birth Cohort (TEBC) study, a world-first study monitoring the progress of 400 babies from birth to adulthood. By monitoring both premature and full-term children’s health, development and educational attainment, researchers are making discoveries about the factors that shape brain development, new treatments for early brain injury, and how childhood experiences contribute to common adult diseases.
Since 2004, the research laboratory has worked to improve understanding of what causes early labour, how we can develop treatments to prevent it and how we can better help newborn babies in the first crucial hours and days after birth.
Video
A world-first study, the Theirworld Edinburgh Birth Cohort, was launched at the Jennifer Brown Research Laboratory in 2015. Researchers from the Laboratory monitor the progress of 400 babies, both premature and full term, from birth to adulthood. Advances in newborn care as a result of the Laboratory’s work are saving lives everyday.
Key discoveries in recent years include:
One of the world’s leading universities, Edinburgh collaborates with prominent institutions in fields as diverse as e-science, engineering and life and medical sciences.
The MRC Centre for Reproductive Health is the only research centre funded by the Medical Research Council (MRC) that focuses exclusively on reproductive health.
NHS Lothian is the UK’s second-largest Health Authority and hosts the Simpson Centre for Reproductive Health at the Royal Infirmary of Edinburgh.